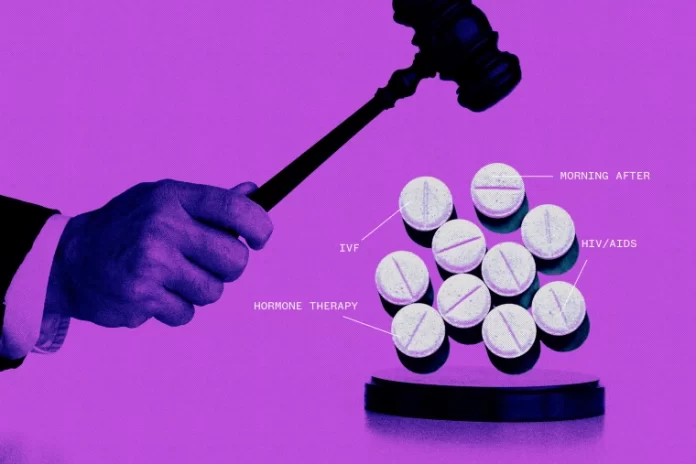
Experts in the pharmaceutical industry caution that if the Supreme Court supports a challenge to the abortion pill mifepristone, it could open the door to legal challenges against vaccines, birth control pills, hormone therapies, and fertility drugs. This challenge isn’t just about access to mifepristone but about potentially undermining the authority of the FDA and causing chaos in the pharmaceutical sector.
The concern is that allowing individual federal judges to disregard the FDA’s scientific findings on health and safety could lead to litigation over various drugs, both existing and those awaiting approval. This precedent might empower anyone with ideological objections to challenge FDA decisions, potentially impacting public health and safety.
Groups representing pharmaceutical companies, including PhRMA, have expressed support for the FDA’s authority. They worry that a ruling against the government could deter investment in the industry, hindering innovation and ultimately harming patients.
The case also raises concerns about the broader implications for drug regulation. Investors rely on FDA approval as a signal of a drug’s safety and efficacy. If FDA decisions are constantly subjected to legal challenges, investors may become more cautious, affecting the development and availability of new treatments.
While the legal dispute centers on the FDA’s handling of mifepristone, its outcome could have implications for other drugs and the FDA’s regulatory authority. The case highlights tensions between abortion rights and FDA regulations, with conservative justices likely to play a significant role in shaping the outcome.
Pharmaceutical industry experts are warning of potential legal ramifications across various medications if the Supreme Court backs a challenge to the abortion pill mifepristone. This challenge doesn’t just threaten access to mifepristone but also questions the FDA’s established regulatory processes, raising concerns about broader implications for drug approval and litigation.
The fear is that if individual federal judges are empowered to overturn FDA findings based on personal beliefs rather than scientific evidence, it could trigger lawsuits against a range of medications. This includes vaccines, birth control pills, hormone therapies, and fertility drugs, sparking uncertainty and disruption in the pharmaceutical sector.
Support for the FDA’s authority comes from pharmaceutical executives and industry groups like PhRMA, emphasizing the importance of regulatory stability for innovation and investment. They argue that a ruling against the government could deter investors, potentially stifling research and development efforts.
Beyond the immediate implications for mifepristone, the case underscores larger questions about the balance between FDA oversight and individual rights. Conservative justices, who hold a majority on the court, are likely to shape the outcome, with potential ramifications for both abortion rights and drug regulation.